 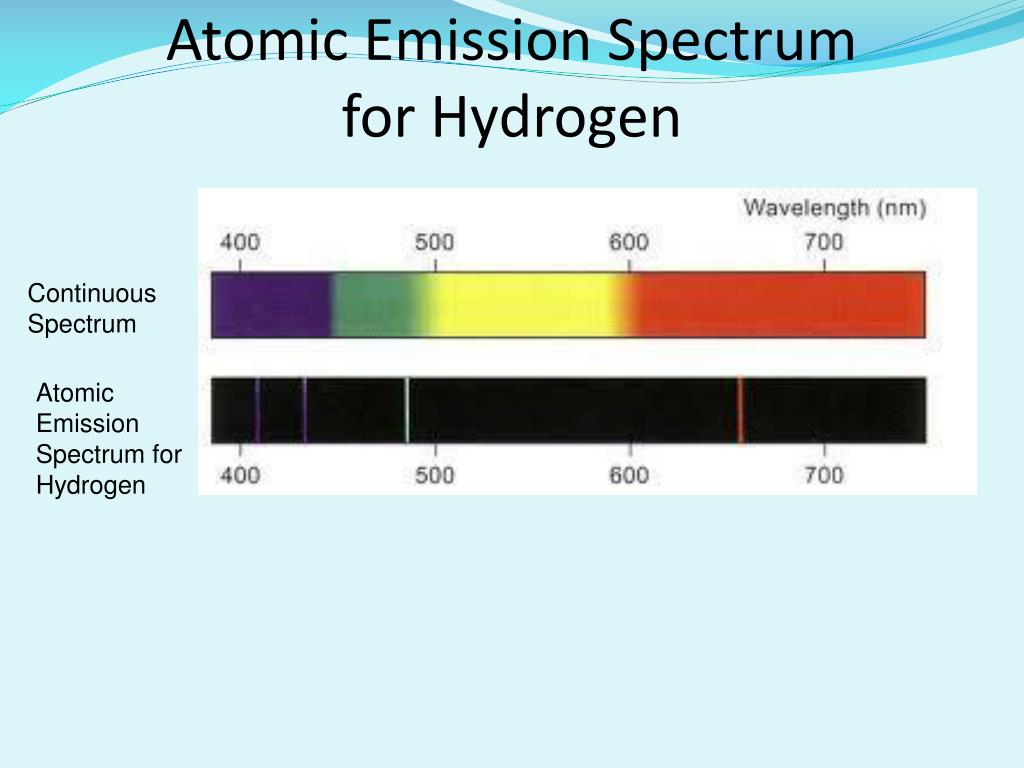 
Consequently, the n = 3 to n = 2 transition is the most intense line, producing the characteristic red color of a hydrogen discharge (part (a) in Figure 2.3.1 ). At the temperature in the gas discharge tube, more atoms are in the n = 3 than the n ≥ 4 levels. Because a sample of hydrogen contains a large number of atoms, the intensity of the various lines in a line spectrum depends on the number of atoms in each excited state. The n = 3 to n = 2 transition gives rise to the line at 656 nm (red), the n = 4 to n = 2 transition to the line at 486 nm (green), the n = 5 to n = 2 transition to the line at 434 nm (blue), and the n = 6 to n = 2 transition to the line at 410 nm (violet). Thus the hydrogen atoms in the sample have absorbed energy from the electrical discharge and decayed from a higher-energy excited state ( n > 2) to a lower-energy state ( n = 2) by emitting a photon of electromagnetic radiation whose energy corresponds exactly to the difference in energy between the two states (part (a) in Figure 2.11 ). As shown in part (b) in Figure 2.11, the lines in this series correspond to transitions from higher-energy orbits ( n > 2) to the second orbit ( n = 2). We can now understand the physical basis for the Balmer series of lines in the emission spectrum of hydrogen (part (b) in Figure 2.9 ). Bohr calculated the value of \(\Re\) from fundamental constants such as the charge and mass of the electron and Planck's constate and obtained a value of 1.0974 × 10 7 m −1, the same number Rydberg had obtained by analyzing the emission spectra. The negative sign in Equation 2.11 and Equation 2.12 indicates that energy is released as the electron moves from orbit n 2 to orbit n 1 because orbit n 2 is at a higher energy than orbit n 1. \)Įxcept for the negative sign, this is the same equation that Rydberg obtained experimentally. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
